FLUID BIOMARKERS RESEARCH STREAM
A biomarker, or biological marker, is a measurable indicator of a disease. AIBL researchers use different types of biomarkers in combination with demographic, clinical and lifestyle data to better understand the nature of Alzheimer’s disease, measure its prevalence and progression within the study cohort, and develop tools, such as neuroimaging markers or screening tests.
Research method
At the beginning of the study, every AIBL study participant provides an 80 mL blood sample and (optionally) cerebrospinal fluid (CSF) samples.

A portion of the blood sample is used in a range of baseline testing, from clinical pathology screening to novel biomarker examinations. 0.5 mL of whole blood is forwarded for apolipoprotein E genotyping, and another 2 mL of whole blood is stored in liquid nitrogen. The remaining blood is fractionated into serum, plasma, platelets, red blood cells and white blood cells (stored in dH20 or RNA later) and stored in liquid nitrogen.
Stored blood samples are sourced from three different tube types: EDTA tubes with added prostaglandin E1 (Sapphire Biosciences, 33.3 ng/ml), lithium-heparin tubes, and serum tubes.
Research highlights
- Researchers have developed blood biomarker panels that are diagnostic of disease and prognostic for brain beta-amyloid (Aβ) and tau levels, indicating a likelihood of disease progression. With further improvement, these studies are paving the way for developing a blood-based first-step screening test for Alzheimer’s disease.
- In collaboration with the CRC for Mental Health, researchers have identified, via 2D gel electrophoresis, biomarkers in plasma that distinguish between Alzheimer’s disease and control subjects. As the biomarkers reflect the Aβ load in the brain, they have the potential to detect Alzheimer’s disease at a preclinical stage of the condition. Further validation studies are currently in progress.
- Researchers from The University of Melbourne and AIBL have developed a method of
- screening patients at risk of Alzheimer’s disease. This method detects the expression levels of selected microRNA extracted from small blood samples. This novel method provides a sensitive means of diagnosing Alzheimer’s disease at an early stage (87% sensitivity; 77% specificity). The method has been validated in clinical samples from patients in the AIBL study.
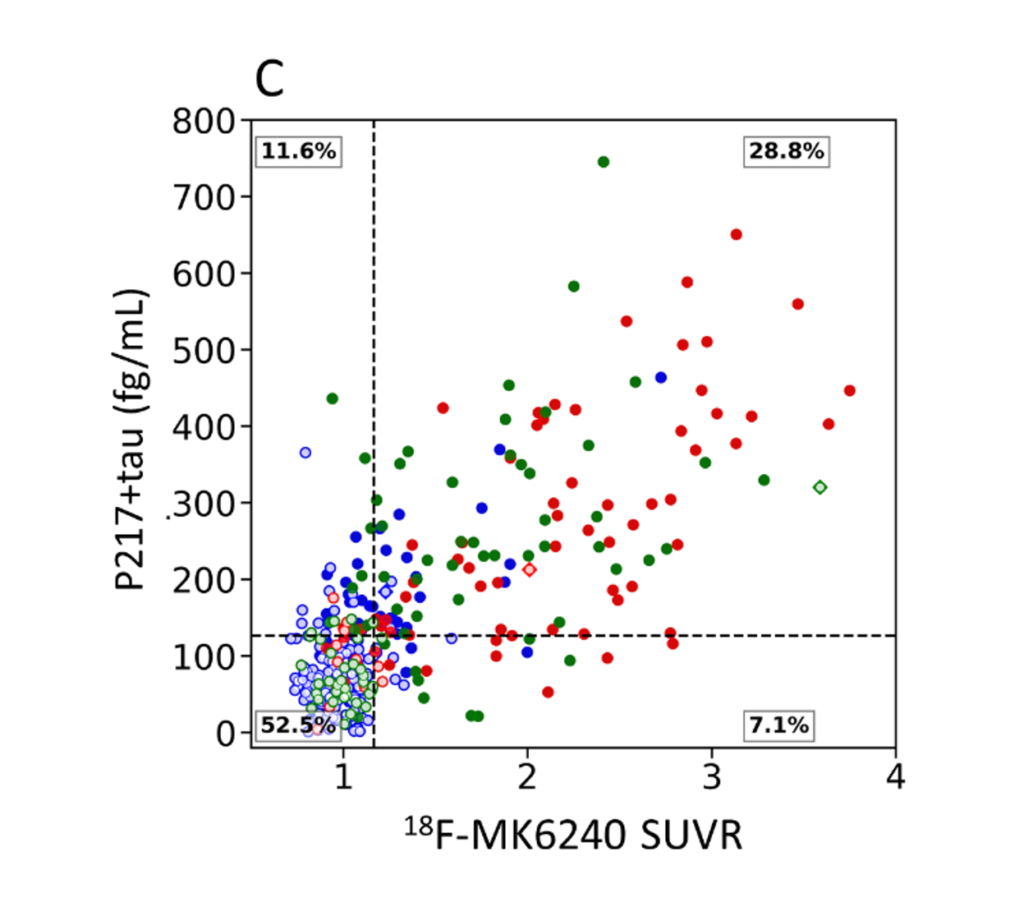
Figure 1: Comparison of a blood biomarker (plasma p217+tau) and PET SUVR measures of tau. Clinical groups are colour-coded with red for dementia, green for mild cognitive impairment and blue for cognitively unimpaired.
LIFESTYLE RESEARCH STREAM
One of the unique aspects of the AIBL study, compared to other international large prospective longitudinal studies of ageing and Alzheimer’s disease, is its dedicated lifestyle research program. The primary aim of the lifestyle program is to examine lifestyle and dietary factors associated with healthy ageing and Alzheimer’s disease.
Research method
AIBL participants complete a series of questionnaires to examine their diet, physical activity patterns and sleep habits. Objective measures of physical activity and body composition are also obtained from a subset of the cohort using ActiGraph Uni-axial Accelerometers and Dual Energy X-Ray Absorptiometry (DXA) scans.

Research highlights
- Identified that older adults who participate in higher levels of physical activity have enhanced cognitive performance and lower levels of Aβ and tau, the brain proteins implicated in Alzheimer’s disease.
- Identified that older adults undertaking higher levels of physical activity have larger brain volumes.
- Found that greater adherence to a Mediterranean diet in older adulthood is associated with less cognitive decline and less accumulation of brain Aβ over time.
- Found that adherence to a Western diet and increased carbohydrate intake is associated with increased cognitive decline over time.
- Found that higher coffee consumption is associated with less cognitive decline and less accumulation of brain Aβ over time.
- Identified that prolonged time taken to fall asleep is associated with greater brain Aβ.
- Showed that lifestyle may play a role in mitigating an increased genetic risk of Alzheimer’s disease.
neuroimaging RESEARCH STREAM
The AIBL study was the first major observational study of ageing and Alzheimer’s disease to extensively use amyloid PET scans. It remains one of the largest global studies involving Positron Emission Tomography (PET) scans for beta-amyloid (Aβ) and tau measurement. This technology is considered the gold standard for imaging brain Aβ and tau in Alzheimer’s disease (Pilebro et al. 2018).
Research method
Participants of the AIBL study undergo Magnetic Resonance and several PET scans.
Aß PET scans
Aβ PET imaging has been used in the AIBL study since it began in 2006. This technology has provided a reliable and accurate measure of beta-amyloid (Aβ) burden in the brain and tremendously improved researchers’ understanding of Alzheimer’s disease. PET imaging involves intravenous injection of a small amount of radioactive tracer that binds to Aβ plaques. In the early years of AIBL, researchers used 11C-PIB marker, the benchmark radioactive marker for Aβ imaging. However, due to the short radioactive decay half-life of 11C, longer-lasting Fluorine-18 labelled tracers were developed for amyloid PET, including 18F-Florbetaben, 18F-Florbetapir, 18F-Flutemetamol and 18F-NAV4694. 18F-NAV4694 can detect lower levels of Aβ in the early stages of Alzheimer’s disease and shows a better binding profile relative to PIB. It is now the primary tracer used in AIBL, despite no FDA approval.
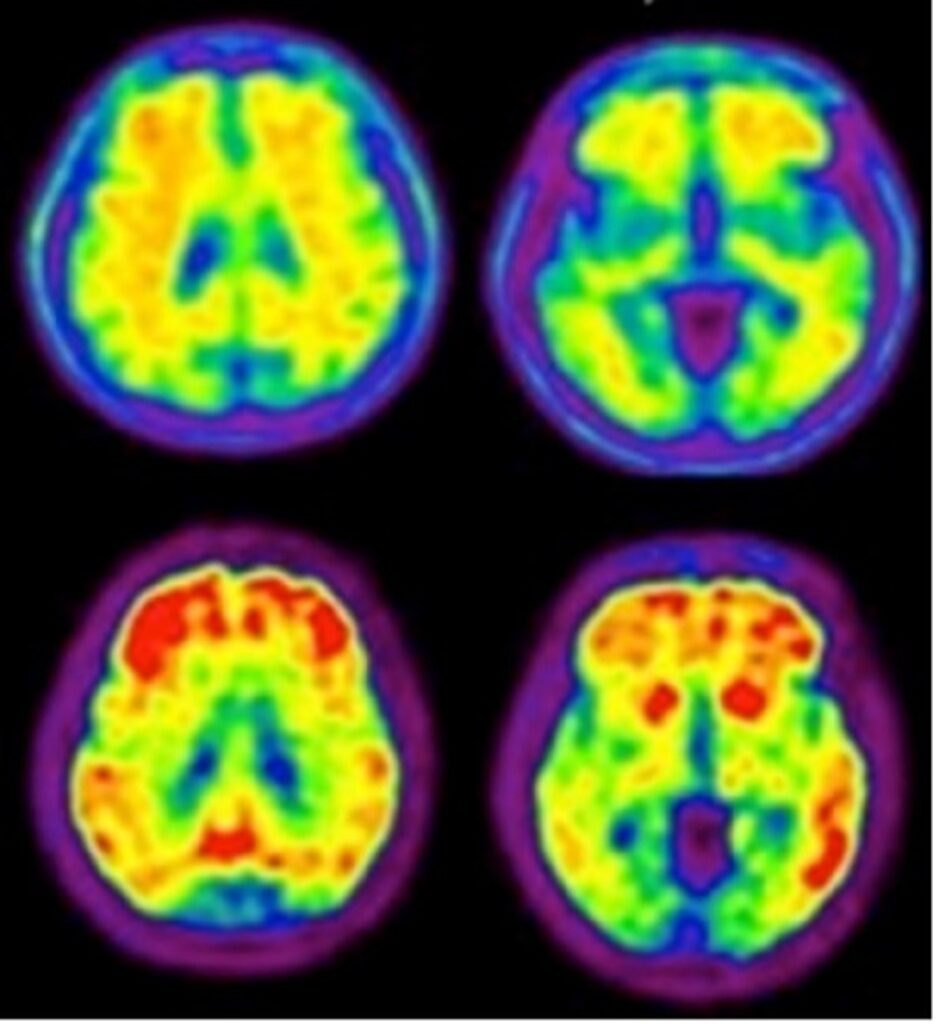
Figure 1: Same patient comparison of an FDA-approved 18F-Aβ tracer (top row) to 18F-NAV4694 (bottom row) (Krishnadas 2021).
Tau PET scans
Aβ imaging can be used to rule out the presence of Aβ in the brain—an indicator of Alzheimer’s disease as a cause of cognitive impairment. However, the high prevalence of Aβ in normal, older individuals limits the use of Aβ PET as a ‘rule-in’ diagnostic test for Alzheimer’s disease.
Tau neurofibrillary tangles are another hallmark of Alzheimer’s disease. Recent studies show that a high level of tau is more closely associated with the onset of cognitive decline than Aβ. This makes it a valuable addition to the imaging biomarkers, especially to confirm a diagnosis of Alzheimer’s disease.
AIBL has been a leader in developing a second-generation 18F-MK6240, which gives higher contrast images with lower non-specific and off-target binding. It has been the preferred tau tracer for the AIBL cohort since 2018.
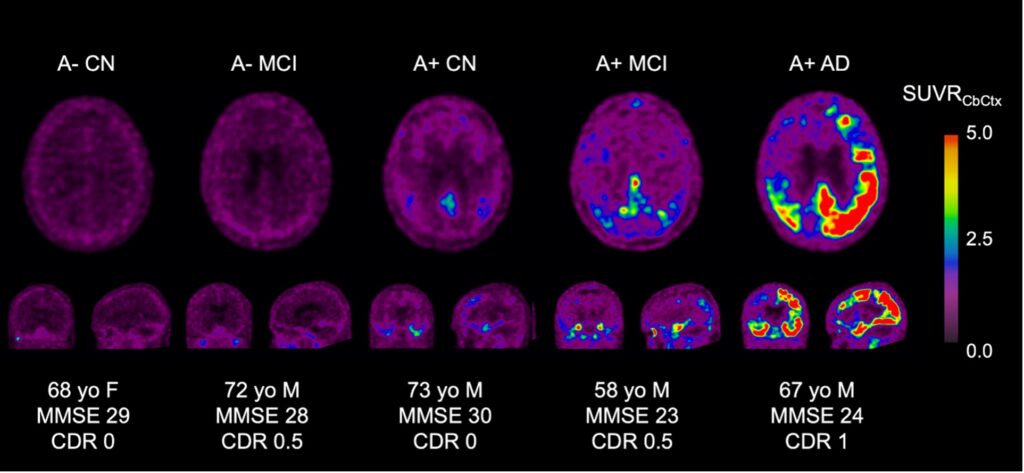
Figure 2: Representative axial, coronal and sagittal tau PET images across the spectrum of Alzheimer’s disease.
Mao-B PET scans
Aβ plaques and neurofibrillary tangles of tau protein are accompanied by reactive gliosis, cellular degeneration and diffuse synaptic and neuronal loss.
Reactive gliosis, characterised by reactive astrocytes and activated microglia, contributes greatly to neurodegeneration throughout the course of Alzheimer’s disease. Reactive astrocytes overexpress monoamine oxidase B (MAO-B).
AIBL is using a PET tracer called 18F-SMBT-1, a highly selective MAO-B tracer, to study astrogliosis. 18F-SMBT-1 will allow a better understanding of the pathophysiology of Alzheimer’s disease while examining its potential direct or indirect effect over neurodegeneration, cognitive decline and clinical progression. This will enable more accurate staging and prognosis at earlier stages of the disease.
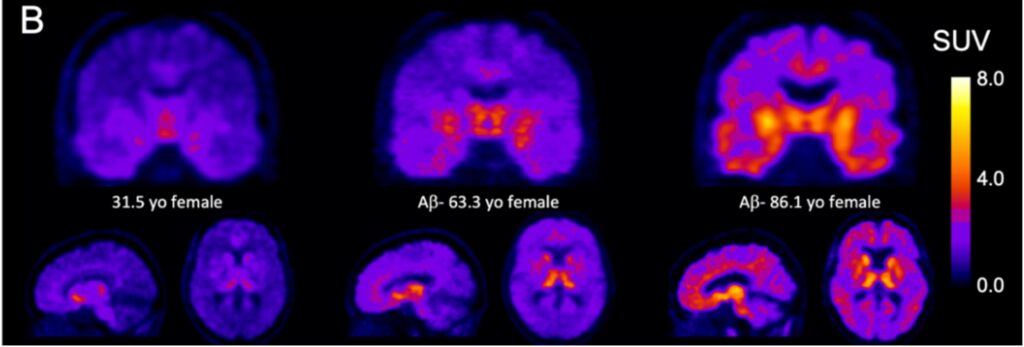
Figure 3: Coronal, sagittal and transaxial images illustrating the age-related regional increases in 18F-SMBT-1 signal in three females aged 31 to 86.
MR imaging
MRI is a powerful method to capture detailed anatomical images of different brain tissues. MRI is an essential tool, providing an assessment of anatomical characterisation and degeneration (T1W, PDW, T2W), the structural integrity of white matter (DWI), perfusion (ASL) and pathological imaging (FLAIR, SWI). In particular, T1W acquisitions offer excellent contrast between white and grey matter, allowing the precise delineation of the outer cortical mantle. This allows the degree of neuronal (grey matter) loss to be accurately measured.
Research highlights
- Researchers have found that PiB-PET scans can detect abnormal levels of beta-amyloid (Aβ) in the brain 15 years before the typical levels of amyloid are reached in patients with Alzheimer’s disease dementia. The total accumulation time for Aβ from zero to typical amounts found in people with Alzheimer’s disease occurs over 25–30 years.
- Researchers have determined that hippocampal atrophy becomes detectable using MRI four years before the onset of dementia.
- Researchers have demonstrated that the level of Aβ provides important prognostic information on a person’s risk of developing dementia and should assist the design of therapeutic trials in preclinical Alzheimer’s disease (Van der Kall et al. 2021).
- Researchers have determined that hippocampal atrophy becomes detectable using MRI four years before the onset of dementia.
- Researchers have also determined that memory impairment becomes abnormal on neuropsychology tests three years before the onset of dementia.
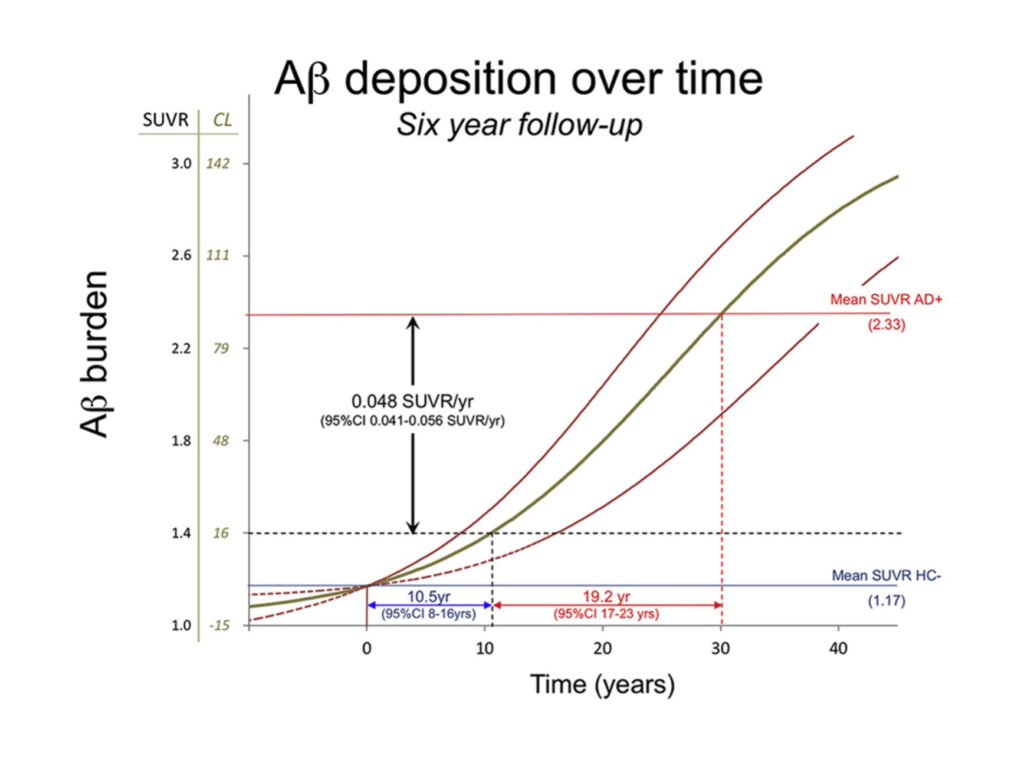
Figure 4: The natural history of Aβ deposition in sporadic Alzheimer’s disease.
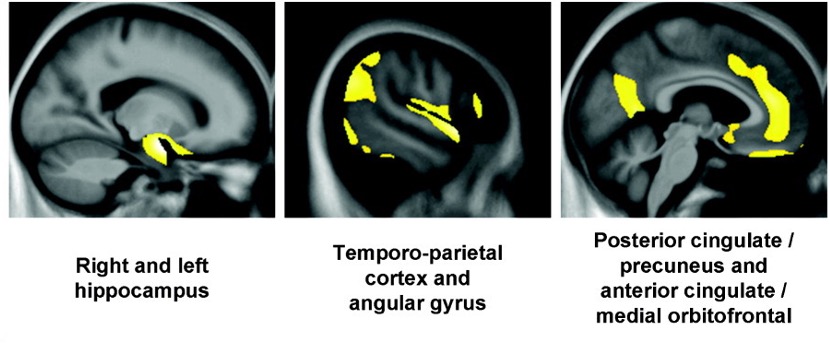
Figure 5: Brain areas of significant negative correlation between Ab and grey matter volume in a cognitively unimpaired individual with a high level of Ab in their brain.
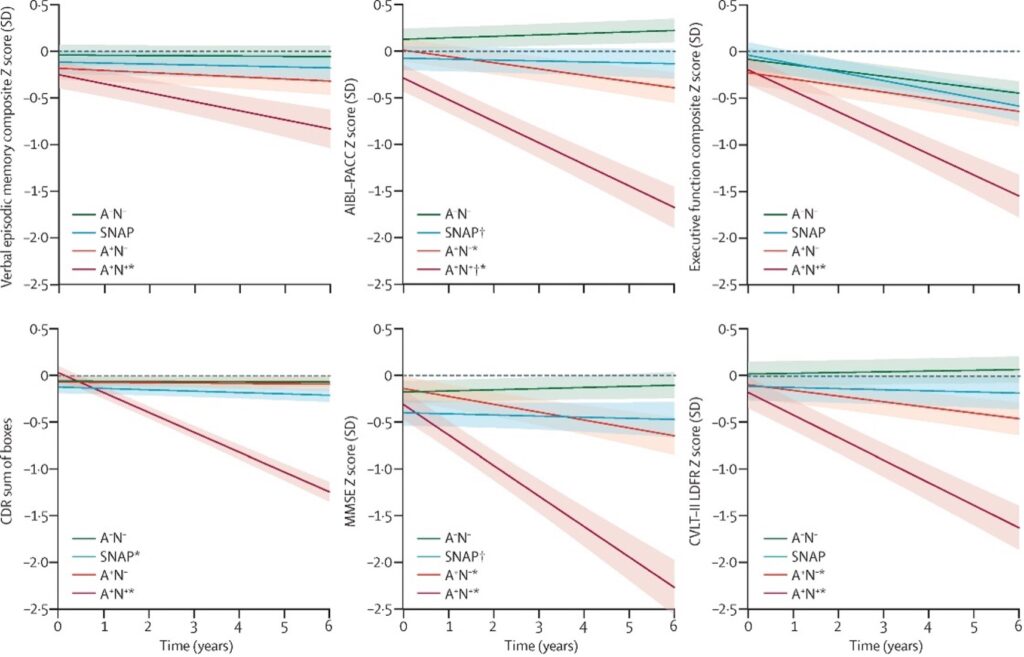
Figure 6: Cognitive trajectories of cognitively unimpaired individuals without Ab and neurodegeneration (green), without Ab and with neurodegeneration (blue), with Ab and without neurodegeneration (orange) and with Ab and neurodegeneration (red).
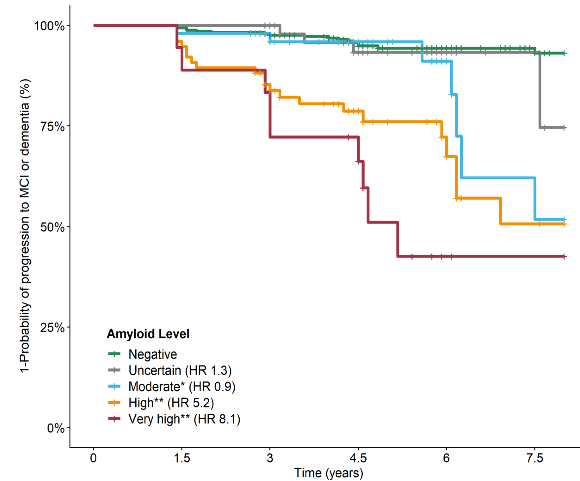
Figure 7: Kaplan-Meier survival analysis by Ab level in normal, older persons. An event was defined as progression to mild cognitive impairment or dementia.
CLINIcal and cognitive RESEARCH STREAM
AIBL researchers conduct cognitive testing to assess cognitive functioning and establish the severity of cognitive impairments.
Research method
At each visit, AIBL participants complete a comprehensive set of assessments and rating scales. Each test and scale is administered and analysed by trained psychologists and neuropsychologists, with the entire assessment requiring between one and two hours, with breaks.

The tests and scales given in each AIBL assessment are selected based on:
- their established validity in the neuropsychology of Alzheimer’s disease
- their use in clinical trials of Alzheimer’s disease
- their ability to be given repeatedly.
At each visit, detailed data is collected on the memory, executive function and attention of every AIBL participant. Participants also undergo a detailed clinical interview using the Clinical Dementia Rating Scale (CDR) scale, Mini Mental Status Examination and ratings of depression and anxiety. They also complete a detailed questionnaire about their family medical history (including a history of psychiatric disorders, dementia and other neurological illnesses), personal medical history, medication use and smoking, and questions about current and past alcohol and illicit drug use.
After each assessment, a consensus panel examines all the data for each participant and classifies their clinical disease stage (e.g., cognitively unimpaired, mild cognitive impairment, dementia).
Together, this clinical and cognitive data provide a sound basis for:
- identifying, classifying and staging Alzheimer’s disease.
- characterising how cognition and mood can change with normal ageing and in early and symptomatic Alzheimer’s disease.
- developing brain-behaviour models of the disease.
Research highlights
- Neuropsychological data confirmed that abnormally high levels of beta-amyloid (Aβ), detected using PET, is associated with subtle but relentless cognitive decline in individuals with dementia and mild cognitive impairment, and most importantly, cognitively unimpaired older adults. These findings contributed to the definition of the preclinical stage of Alzheimer’s disease.
- While depression and anxiety are exclusion criteria for the AIBL study, data showed that some individuals demonstrated elevated levels of anxiety or depressive symptoms.
- Studies in the AIBL preclinical Alzheimer’s disease sample identified that slight increases in anxiety symptoms are associated with a faster rate of cognitive decline. Furthermore, individuals with increased anxiety show increased blood markers of stress.
- Clinical data from the AIBL study confirmed that developing Alzheimer’s disease dementia requires abnormally high levels of Aβ. Data also confirmed that this elevation in Aβ can be detected up to 30 years before individuals meet any clinical criteria for dementia.
- While cognitively unimpaired adults with abnormally high levels of Aβ show progressive decline in cognition, mainly in memory, data showed there is variability between these individuals in the rates of Aβ -related cognitive decline. This suggests other factors could influence the biology of Alzheimer’s disease. Analysis of neuropsychological data from the AIBL sample shows that allelic variability on a common gene associated with the growth of brain cells and their interconnections with others (Brain Derived Neurotrophic Factor (BDNF)) enabled some older adults to withstand the damaging effects of increasing Aβ on their memory.
- Researchers have found that the learning impairment in the preclinical Alzheimer’s disease group is more than three times greater than impairments detected from 10 years of following memory decline in the same people. This data suggests that in very early Alzheimer’s disease, the main cognitive impairment is a failure to benefit from experience, rather than in the ability to remember previously learned information.
- Continual cognitive and clinical assessment and categorisation of the AIBL cohort is the foundation for all research conducted within AIBL. David Ames personally reviewed all AIBL participant files over five different time points. These files are subsequently reviewed for cognitive data accuracy by the neuropsychology team at The Florey Institute. This attention to detail by a leading clinician in the field of Alzheimer’s disease research has resulted in the high diagnostic accuracy within the AIBL study, setting it apart from many other large-scale cohort studies.
Genetics RESEARCH STREAM
Genetics and epigenetics have an important influence on our risk of developing Alzheimer’s disease and how the disease progresses after its onset. Genetic factors can include differences in the actual DNA sequence; and markers on top of the DNA sequence, known as epigenetic markers or DNA methylation. Alzheimer’s disease risk can be attributed to single genetic or epigenetic factors or combinations of factors in the form of scores and profiles.
Research method
Genetic and epigenetic information is studied alongside other clinical and biological data within the AIBL study to determine which factors best predict Alzheimer’s disease and how it progresses. The results from these investigations can deliver scores that can help identify individuals at most risk of Alzheimer’s disease. They can also help identify biological processes that could be important in developing Alzheimer’s disease.

Genetic and epigenetic information is studied alongside other clinical and biological data within the AIBL study to determine which factors best predict Alzheimer’s disease and how it progresses. The results from these investigations can deliver scores that can help identify individuals at most risk of Alzheimer’s disease. They can also help identify biological processes that could be important in developing Alzheimer’s disease.
DNA sampling
All AIBL participants provide 5 ml whole blood samples for DNA extraction using QIAamp DNA Blood Maxi Kits (Qiagen). DNA is aliquoted and stored at neat or 100 ng/μL concentrations at –80◦C for future use.
APOE genotyping
The single greatest genetic risk factor for Alzheimer’s disease is found within the APOE gene, encoding the Apolipoprotein E protein. The genotype for this genetic factor presents as any combination of two of the following: ε2, ε3 and ε4. People with one (ε2/ε4 or ε3/ε4) or two (ε4/ε4) copies of the ε4 genotype have a significantly increased risk of Alzheimer’s disease. This genotype is defined using two genetic variants or single nucleotide polymorphisms, specifically rs7412 and rs429358. This is determined using Taq Man® genotyping assays on a Quant-StudioTM 12K Flex Real-Time-PCR system (Applied Biosystems).
Genome-wide SNP genotyping
Genome-wide single nucleotide polymorphism (SNP) array data is collected using the Axiom Precision Medicine Diversity Array (Applied Biosystems™). This data has been collected from over 2000 AIBL participants. A further subset of 1357 early study participants has undertaken genome-wide analysis using the OmniExpressHumanExome + BeadChip (Illumina). All genetic datasets are imputed using both the TopMed and HRC Imputation Servers.
Whole-exome sequencing
Whole-exome sequencing has been undertaken on a subset of 497 AIBL participants using 1 μg of DNA, extracted and stored as described above. The SureSelect Human All Exon V5 (51 Mb) Kit (Agilent Technologies) was used for exonic sequence enrichment. The resulting libraries were sequenced on the Illumina HiSeq2500 using 100 bp paired-end read chemistry. The Burrows-Wheeler Aligner (BWA) was employed for sequence alignment to the human genome reference (build GRCh37).
Genome-wide DNA methylation quantification
Genome-wide DNA methylation data is available for 726 AIBL participants at a single time point. Additionally, for 400 of these participants, two additional time points of DNA methylation data were generated. Time points for DNA methylation estimation were selected based on the best alignment with available neuroimaging and clinical data. DNA samples, as outlined above, were processed using Amicon® Ultra-0.5 Centrifugal Filters (Merck) for the removal of guanidine isothiocyanate introduced during DNA extraction, and 500 ng of resultant DNA was bisulphite, converted using EZ DNA Methylation Kits (ZYMO Research). The HumanMethylationEPIC BeadChip Array (Illumina) was used for the determination of genome-wide DNA methylation.
Research highlights
- In collaboration with the neuroimaging and cognitive research streams, researchers have increased understanding of the role individual genes, specifically APOE, BDNF, KIBRA and SPON1, have in influencing cognitive decline during the preclinical and prodromal stages of Alzheimer’s disease.
- Researchers have developed novel genetic profiles and scores to assess the cumulative effect of genetic variants on cognitive decline, Aβ PET markers and brain volumetrics (cumulative genetic risk). These scores have been developed in the presence and absence of the largest genetic risk factor, APOE.
- In collaboration with the lifestyle stream, the genetics stream has driven research into understanding the impact of gene-lifestyle interactions in Alzheimer’s disease. Researchers have explored the interaction of genetics with physical activity, diet and sleep by focusing on established Alzheimer’s disease genes (e.g. APOE and BDNF), accumulative genetic risk (polygenic risk scores) and both lifestyle a priori (e.g. AQP4) and unbiased (discovery) approaches.
- Researchers have assessed epigenetic clocks and disproportionate biological ageing to reveal associations with hippocampal volume.
DATA and machine learning RESEARCH STREAM
Data collected by AIBL and its collaborative partners is assessed to define the underlying nature of Alzheimer’s disease. From blood, CSF and lifestyle to imaging and cognitive data, AIBL researchers use the best statistical methods to answer research questions.
Blood test for Alzheimer’s disease
AIBL scientists have been working to devise the most appropriate markers for both preclinical and clinical disease diagnosis using a blood test.

Research highlights
- AIBL scientists, in collaboration with a group in Japan, published a paper in the journal, Nature, describing the first blood test to identify Alzheimer’s disease pathology with greater than 90% accuracy.
- Researchers have defined, with high accuracy, the longitudinal nature of Alzheimer’s disease pathology. In this work, scientists mapped the age when pathology begins to grow in the brain and how long it takes before clinical symptoms occur.
- Working with collaborators, AIBL scientists aligned cognitive change with the presence of Alzheimer’s disease pathology and the presence or absence of certain blood markers. A simple blood test and cognitive evaluations can predict the likelihood of cognitive change over the next five years.
Multimorbidity and polypharmacy stream
People with Alzheimer’s disease tend to have more comorbidities and are often prescribed 5-10 more medications than those without Alzheimer’s disease, even after adjusting for age and gender. AIBL researchers will investigate how common comorbidities and the prescribed medications impact on the clinical evolution of Alzheimer’s disease. In addition, the impact of the complex interaction between multimorbidity and Alzheimer’s disease on medication use safety will be explored. We will also develop digital tools to improve precision medication use in people experiencing diseases of ageing.
Research method
AIBL participants complete a comprehensive questionnaire to report their medical history and medication use. The concentration of the medications in the blood will be assessed using our high throughput assays.

In conjunction with demographic data and other clinical assessment performed in AIBL study, epidemiological, biostatistical and innovative machine learning analysis will be employed to achieve our research goals.
Research highlights
- Anxiety and other neurological disorders are associated with increased likelihood of AD, while arthritis, cancer, gastric complaints, high cholesterol are associated with decreased likelihood of AD.
- Alzheimer’s disease affects how our body handles medication, by altering the expression and function of drug transporters and metabolising enzymes in the intestine, liver and kidney.
- Altered oral absorption of several medications, including valsartan and digoxin, has been identified in Alzheimer’s disease.
- Model-informed precision dosing models are under development to promote personalised medicine in people living with Alzheimer’s disease.



View our current substudies
See how the AIBL study supports research in emerging ideas and technologies.


Participate in the AIBL study
Participate in ground-breaking Alzheimer’s disease research.News and events
Get the latest global news about dementia research.
Publications
Explore innovations and clinical developments in Alzheimer’s disease.
Stay up to date with the latest news from the AIBL study.